There are four main categories of medical waste in a dental office: sharps, infectious, hazardous, and non-hazardous. Learn about each here.
As a waste management and compliance company we often get asked by dentists “What are the different types of waste I am required to dispose of and show documentation for?” There are four main types of dental waste, and each comes with its own standard for proper disposal. In this post we will discuss the types of dental waste and their disposal methods:
- Amalgam waste in a dental office
- Hazardous waste in a dental office
- Pharmaceutical waste in a dental office
- Medical Waste in a dental office
Amalgam Waste
Where do you dispose of amalgam scraps is a question often asked.
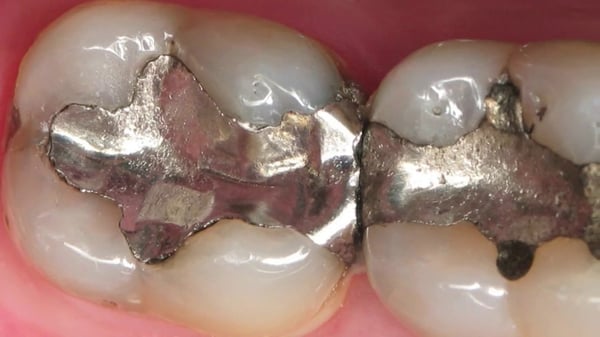
According to the American National Standards Institute/American Dental Association (ANSI/ADA), amalgam waste is defined as: “including amalgam (scrap), chair-side trap filters containing amalgam vacuum pump filters containing, amalgam, saliva ejectors if used in dental procedures involving amalgam, used amalgam capsules, extracted teeth with amalgam restorations, and waste items that are contaminated with amalgam”. (1) If not disposed of properly, amalgam waste runs the risk of being introduced to the waterways and environment, and impacting a community. The most effective ways to prevent this amalgam waste from entering the environment are through the use of amalgam separators and an amalgam recycling program. As of 2017, the EPA has ruled that amalgam separators must achieve at least a 95% removal efficiency. (2) That separated amalgam waste cannot then be discarded in the trash. Instead, it must be sent to a facility that specializes in melting the metals and recycling that mercury.
Hazardous Waste

Hazardous waste is defined as waste that has “substantial or potential threats to public health or the environment”. To be classified as hazardous, the waste must exhibit one of the following traits: Ignitability, Reactivity, Corrosivity, or Toxicity. (3) Hazardous waste can be found in any physical state; liquid, gaseous, or solid. The type of disposal depends entirely upon the physical state the waste is in, and determines the need for further treatment. To prevent the introduction of hazardous waste into the environment, countermeasures such as solidification or the erection of barriers to contain the waste may be necessary. One thing is certain: hazardous waste cannot be simply thrown away in the garbage or washed down the sink. Each state differently regulates the disposal of hazardous waste, a map of which you can find HERE.
Pharmaceutical Waste
![]()
The EPA defines pharmaceutical waste as expired, unwanted or unused pharmaceuticals. “Healthcare facilities and healthcare-related businesses that generate pharmaceutical wastes are responsible for appropriately managing their wastes in accordance with all local, state and federal environmental regulations.” (4) This management can come in the forms of take-back programs or incineration. Mail-back systems exist as a convenient and inexpensive option to dispose of the collected pharmaceutical waste.
PureWay disposes of pharmaceutical waste in healthcare facilities and dental facilities. Visit our pharmaceutical waste disposal resources for more details on this service.
Medical Waste
Regulated medical waste can be broken down into two subcategories; Sharps & Biohazard waste.
Biohazardous Waste
Regulated Waste (5) means liquid or semi-liquid blood or other potentially infectious materials; contaminated items that would release blood or other potentially infectious materials in a liquid or semi-liquid state if compressed; items that are caked with dried blood or other potentially infectious materials and are capable of releasing these materials during handling; contaminated sharps; and pathological and microbiological wastes containing blood or other potentially infectious materials.
Sharps waste

A medical sharp is any device or object used to puncture or lacerate the skin. These include common items used in dental offices such as: cap needles, disposable scalpel brackets, wires, and contaminated glass. (6) Once used in this manner, a sharp is now classified as regulated medical waste and must be disposed of properly in a sharps container. Sharps mail-back systems enable any healthcare facility or self-injector to safely dispose of and recycle sharps waste.





